Discovery of UCB9386: A Potent, Selective, and Brain-Penetrant Nuak1 Inhibitor Suitable for In Vivo Pharmacological Studies.
Poullennec, K.G., Jnoff, E., Abendroth, J., Bhuma, N., Calmiano, M., Calmus, L., Cardenas, A., Courade, J.P., Delatour, C., Hall, A., de Haro, T., Delker, S.L., Demaude, T., Gaikwad, N., Ghavate, D., Gholap, A.R., Kierkowicz, M., Le Mestre, R., Van Hijfte, N., Verheijden, S., Vernerova, K., De Wever, V., Waghmode, N.(2024) J Med Chem 67: 20879-20910
- PubMed: 39588908
- DOI: https://doi.org/10.1021/acs.jmedchem.4c01237
- Primary Citation of Related Structures:
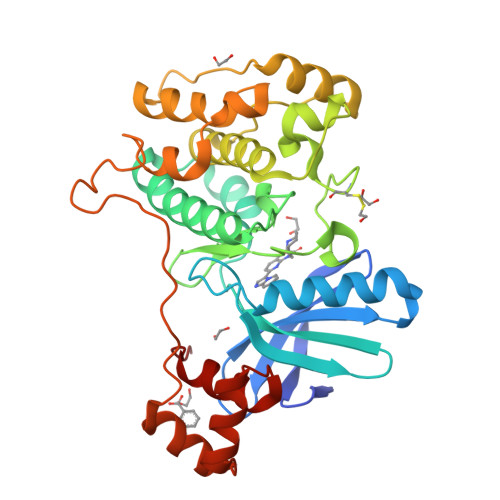
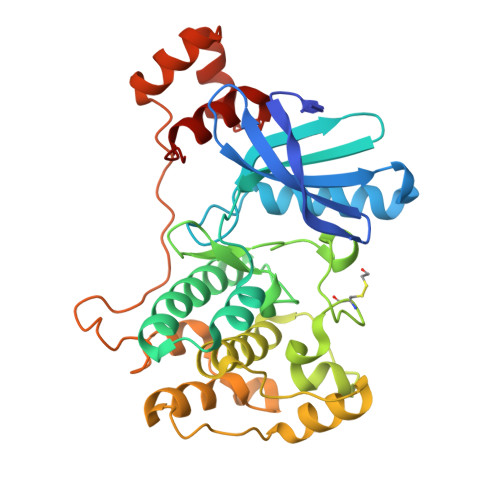
8UOJ, 8UOK - PubMed Abstract:
Nuak1 (NUAK family SnF1-like kinase-1) is a serine-threonine kinase and a member of the AMPK family. Interest in Nuak1 has increased over the years due to the role it plays in several biological processes, from tumor cell invasion and proliferation to Tau stabilization. Nuak1 is expressed in many cancer cell lines and many reports describe this target as an oncogene, the inhibition of which is hypothesized to be valuable for treating various cancer types including glioma. We report here the discovery of Nuak1 inhibitors originating from HTS hit 9 with excellent selectivity and the subsequent medicinal chemistry optimization program, supported by structural information from the first crystal structures of a Nuak1 chimeric protein which provided insights into the binding modes of our compounds. These efforts yielded a nanomolar cell potent, highly selective and brain penetrant Nuak1 inhibitor UCB9386 ( 56 ) suitable for in vivo pharmacological studies for central nervous system (CNS) disorders.
Organizational Affiliation:
Chemin du Foriest, UCB, 1420 Braine l'Alleud, Belgium.