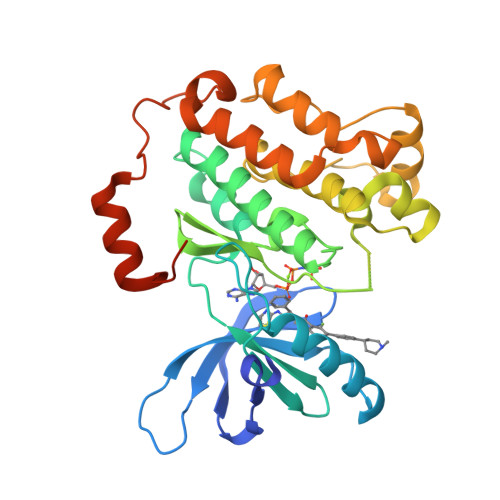
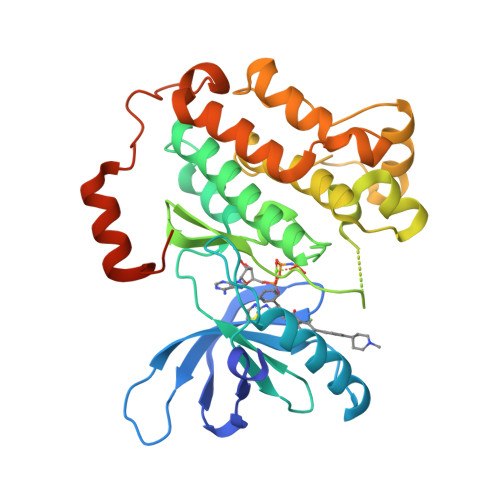
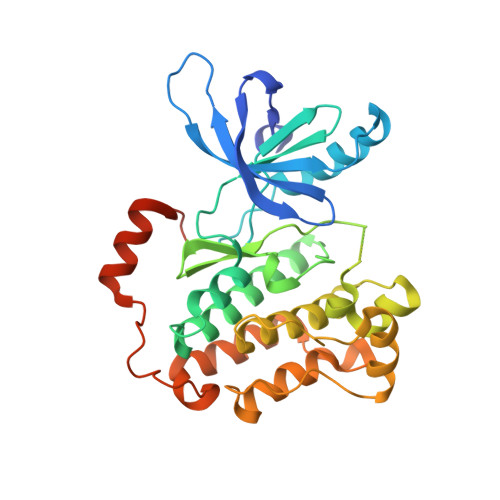
Quinazolinones as allosteric fourth-generation EGFR inhibitors for the treatment of NSCLC.
Gero, T.W., Heppner, D.E., Beyett, T.S., To, C., Azevedo, S.C., Jang, J., Bunnell, T., Feru, F., Li, Z., Shin, B.H., Soroko, K.M., Gokhale, P.C., Gray, N.S., Janne, P.A., Eck, M.J., Scott, D.A.(2022) Bioorg Med Chem Lett 68: 128718-128718
- PubMed: 35378251
- DOI: https://doi.org/10.1016/j.bmcl.2022.128718
- Primary Citation of Related Structures:
7LTX - PubMed Abstract:
The C797S mutation confers resistance to covalent EGFR inhibitors used in the treatment of lung tumors with the activating L858R mutation. Isoindolinones such as JBJ-4-125-02 bind in an allosteric pocket and are active against this mutation, with high selectivity over wild-type EGFR. The most potent examples we developed from that series have a potential chemical instability risk from the combination of the amide and phenol groups. We explored a scaffold hopping approach to identify new series of allosteric EGFR inhibitors that retained good potency in the absence of the phenol group. The 5-F quinazolinone 34 demonstrated tumor regression in an H1975 efficacy model upon once daily oral dosing at 25 mg/kg.
Organizational Affiliation:
Department of Cancer Biology, Dana-Farber Cancer Institute, Boston, MA 02115, USA; Department of Biological Chemistry & Molecular Pharmacology, Harvard Medical School, 360 Longwood Ave, Boston, MA 02115, USA.