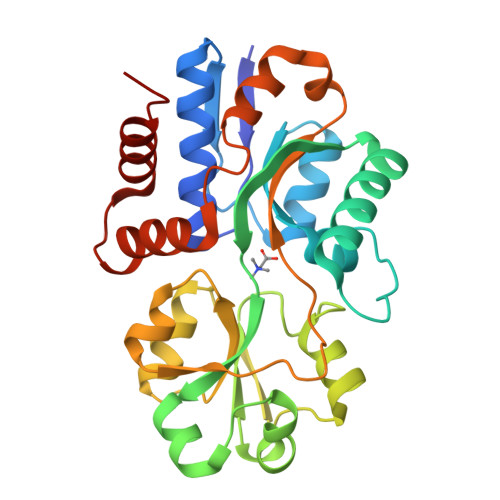
Structural basis for the binding of compatible solutes by ProX from the hyperthermophilic archaeon Archaeoglobus fulgidus.
Schiefner, A., Holtmann, G., Diederichs, K., Welte, W., Bremer, E.(2004) J Biol Chem 279: 48270-48281
- PubMed: 15308642
- DOI: https://doi.org/10.1074/jbc.M403540200
- Primary Citation of Related Structures:
1SW1, 1SW2, 1SW4, 1SW5 - PubMed Abstract:
Compatible solutes such as glycine betaine and proline betaine serve as protein stabilizers because of their preferential exclusion from protein surfaces. To use extracellular sources of this class of compounds as osmo-, cryo-, or thermoprotectants, Bacteria and Archaea have developed high affinity uptake systems of the ATP-binding cassette type. These transport systems require periplasmic- or extracellular-binding proteins that are able to bind the transported substance with high affinity. Therefore, binding proteins that bind compatible solutes have to avoid the exclusion of their ligands within the binding pocket. In the present study we addressed the question to how compatible solutes can be effectively bound by a protein at temperatures around 83 degrees C as this is done by the ligand-binding protein ProX from the hyperthermophilic archaeon Archaeoglobus fulgidus. We solved the structures of ProX without ligand and in complex with both of its natural ligands glycine betaine and proline betaine, as well as in complex with the artificial ligand trimethylammonium. Cation-pi interactions and non-classical hydrogen bonds between four tyrosine residues, a main chain carbonyl oxygen, and the ligand have been identified to be the key determinants in binding the quaternary amines of the three investigated ligands. The comparison of the ligand binding sites of ProX from A. fulgidus and the recently solved structure of ProX from Escherichia coli revealed a very similar solution for the problem of compatible solute binding, although both proteins share only a low degree of sequence identity. The residues involved in ligand binding are functionally equivalent but not conserved in the primary sequence.
Organizational Affiliation:
Fachbereich Biologie, Universität Konstanz, Universitätsstrasse 10, D-78457 Konstanz, Germany. andre.schiefner@uni-konstanz.de